
Concerned about bladder cancer? Cxbladder can give you answers
Cxbladder is a suite of non-invasive genomic tests optimized to help rule out urothelial bladder cancer in patients being monitored for recurrent non-muscle invasive disease, and in those presenting with hematuria (blood in urine).
Cxbladder is an easy-to-use urine test that can reduce the need for invasive procedures when patients are at a low risk of disease.

Genomic testing optimized for the risk stratification of urothelial cancer
Cxbladder is a suite of non-invasive genomic urine tests optimized for the risk stratification of urothelial cancer in patients presenting with hematuria and those being monitored for recurrent disease.
The Cxbladder suite of tests is supported by over 30 peer reviewed publications and Cxbladder Triage is now included in the AUA/SUFU Microhematuria Guideline*.

Optimize your clinical practice while improving the patient experience
The Cxbladder suite helps you prioritize your time and clinical resources on those that need it the most, streamlining practice workflow and increasing overall efficiency.
Cxbladder works for both you and the patient. Spare patients with a low probability of urothelial cancer the discomfort, anxiety and risks of an invasive procedure. With urine-based sampling and the option of in-home sample collection, the Cxbladder suite allows you to optimize your clinical practice while delivering the best patient experience possible.
Cxbladder for patients
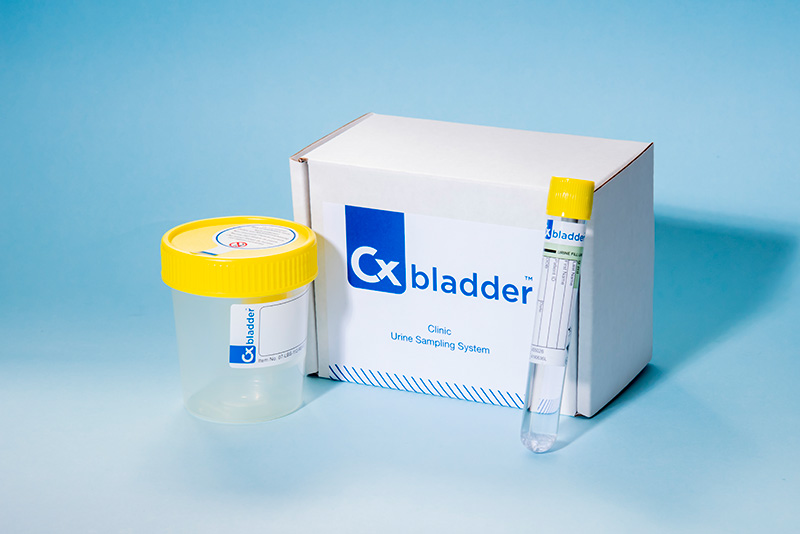
The Cxbladder suite
Learn more about the Cxbladder suite of non-invasive genomic urine tests designed to rule out urothelial bladder cancer, the most common form of the disease.

The sampling system
Learn more about Cxbladder's easy-to-use and non-invasive sampling system, including the option of in-home sampling.
The science
Cxbladder is an advanced laboratory test that analyzes biomarker genes to help rule-out disease.
Latest news
Cxbladder Bulletin - March 2026
Pacific Edge Invites you to read the March 2026 edition of our Cxbladder Bulletin clinician newsletter.
Expert CAC Panel - Recording Now Available
View a recording of the Feb 19 CAC meeting led by Novitas, the MAC with jurisdiction of Pacific Edge’s laboratory operations in Pennsylvania. CACs are typically convened ahead of developing new or substantially revised Medicare policy and the expert panel clearly endorsed the use of urine-based biomarkers in the evaluation of hematuria as medically reasonable and necessary.
First Triage Plus Tests Ordered From Townsville
Pacific Edge announces today that after 11 referrals for Cxbladder were sent to patients last week, the first Triage Plus test has now been ordered by Townsville University Hospital, Queensland, according to an agreed clinical pathway.
Leading Singapore Hospital Now Ordering Cxbladder
Pacific Edge today announces its Dunedin laboratory has signed a service agreement with Singapore General Hospital (SGH), an important milestone in the company’s strategy to drive the adoption of its tests in the Asia Pacific.
References
* Barocas DA, Lotan Y, Matulewicz RS, Raman JD, Westerman ME, Kirkby E, Pak L, Souter L. Updates to Microhematuria: AUA/SUFU Guideline (2025). J Urol. 0(0). doi: 10.1097/JU.0000000000004490.





